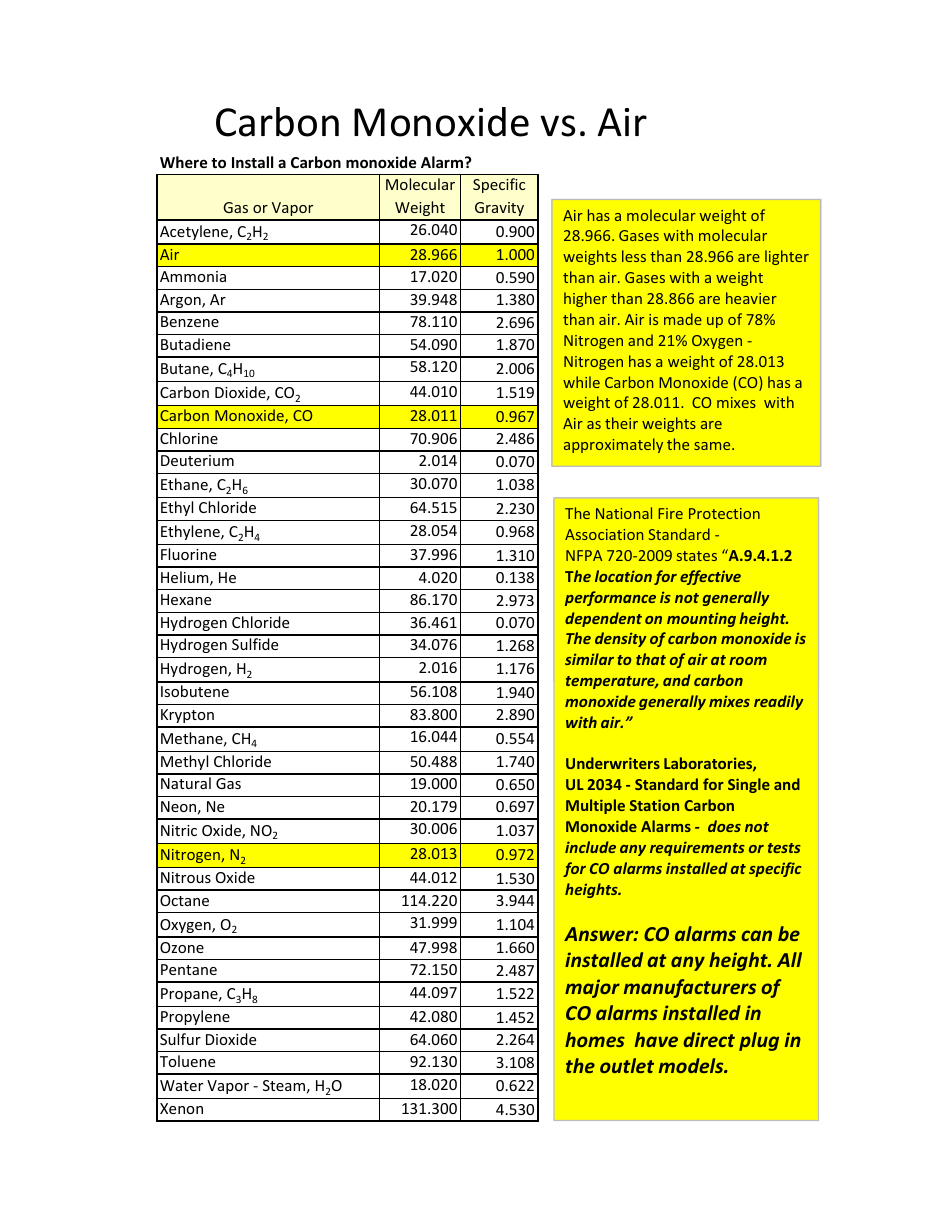
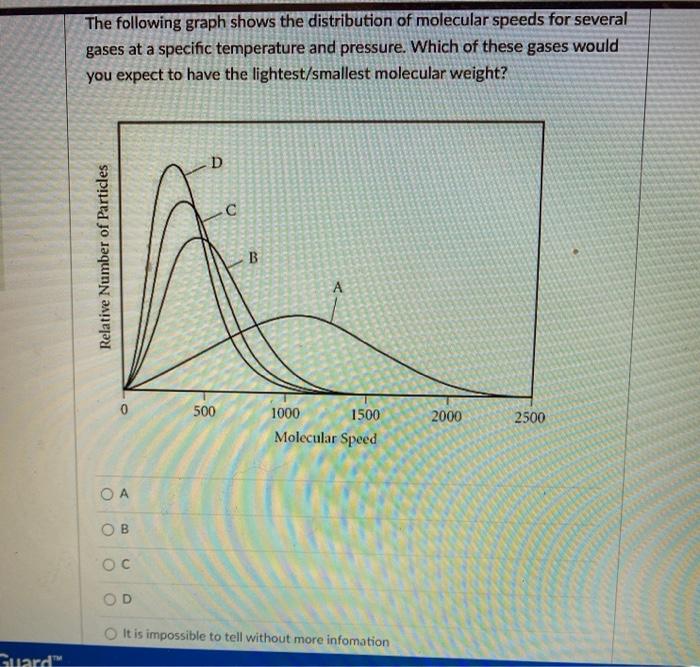
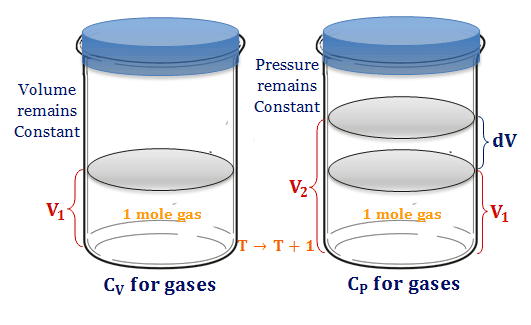
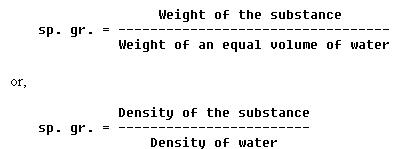SOLVED:the weight per unit volume of a material: the pressure of fluids or gases that are stationary or not in motion. the density of a material divided by the density of' water_

Design of an exhaust gas calorimeter for automobile engines . FIg./O. J3ackFtec&tiUKe: Ca/?d. fft/A/ *2 Fc/l,l. Load. -45* In using the rational equationspreviously mentioned the voume of mixturewas found by multiplying

An Introduction To Density, Specific Gravity and Specific Weight - Formulas With Examples | PDF | Density | Pressure

SOLVED:The van der Waals Equation for real gases is shown below: (P+% (v nb) nRT In the equation, what assumption we made in the formulation of the Kinetic Molecular Theory of gases
Solved] 1) What is the weight of a 5-kg substance in N and lbf? 2) Determine the mass and the weight of the air contained in a room whose dimensions... | Course Hero


![Appendix C: Properties of Gases - Chemical Engineering Fluid Mechanics, 3rd Edition [Book] Appendix C: Properties of Gases - Chemical Engineering Fluid Mechanics, 3rd Edition [Book]](https://www.oreilly.com/library/view/chemical-engineering-fluid/9781498724449/images/tabC_1.jpg)



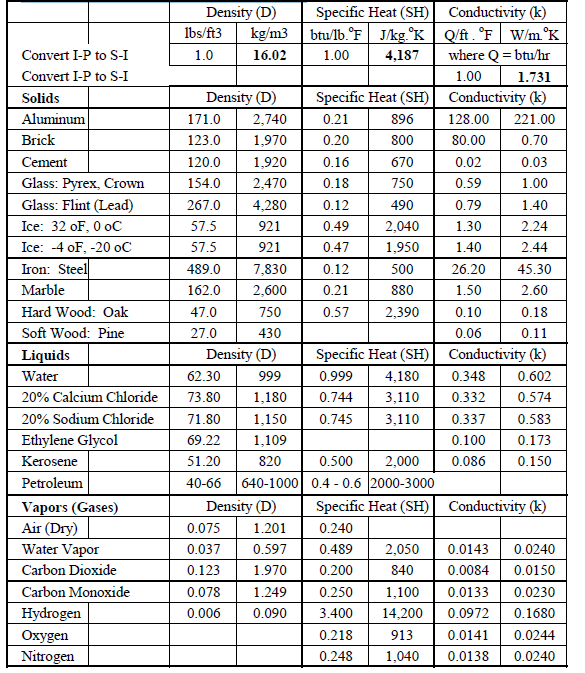

![Solved] Add a new column to Table 10.1, listing the specific gravity of each s | SolutionInn Solved] Add a new column to Table 10.1, listing the specific gravity of each s | SolutionInn](https://www.solutioninn.com/images/question_images/1547/4/5/7/1755c3c5297015c71547439810269.jpg)